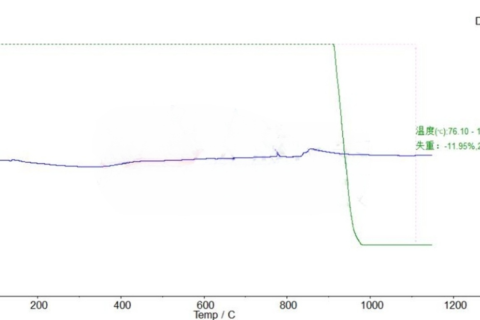
Thermogravimetric Analysis of Lithium Hydroxide Monohydrate (LiOH·H₂O) Using Simultaneous Thermal Analysis (STA)

With the rapid growth of the new energy materials industry, lithium hydroxide monohydrate (LiOH·H₂O) has become a key intermediate in lithium chemical processing. It is widely used in cathode material production, coating additives, lubricating greases, and glass-ceramics.
Understanding its dehydration and decomposition behavior is essential—not only for material purity, but also for optimizing sintering temperatures, storage conditions, and composition control. At CeramXpert, we leverage precise thermal analysis to support reliable material performance in industrial applications.
🔬 Experimental Procedure
- Instrument: Simultaneous Thermal Analyzer (STA)
- Sample: Lithium Hydroxide Monohydrate (LiOH·H₂O)
- Atmosphere: Oxygen
- Heating Rate: 5 °C/min
- Temperature Range: 25 °C – 800 °C
Note: Oxygen atmosphere better simulates real conditions such as oxidation roasting and sintering processes.
📊 Thermal Analysis Results
Stage 1: Dehydration of Crystal Water
- Temperature Range: 31.8 °C – 130.3 °C
- Weight Loss: ≈ 11.31%
- Thermal Effect: Strong endothermic peak (~90 °C)
- Reaction: LiOH·H₂O → LiOH + H₂O
Insight:
Drying temperatures must exceed 130 °C for complete dehydration. Below this, moisture removal is incomplete even over extended storage.
Stage 2: Thermal Decomposition of LiOH
- Temperature Range: 198.9 °C – 456.4 °C
- Weight Loss: ≈ 12.53%
- Thermal Effect: Second endothermic peak (~276 °C)
- Reaction: 2LiOH → Li₂O + H₂O
Insight:
200 °C – 450 °C is the critical decomposition range.
- Sintering within this range requires careful control due to water evaporation.
- Prolonged exposure may lead to lithium loss, stoichiometric deviation, and increased oxygen content.
Stage 3: High-Temperature Stability
- Temperature Range: 590.7 °C – 744.4 °C
- Weight Loss: ≈ 0.32%
Insight:
No significant reactions occur—this is a stable phase region.
✅ Key Conclusions
- Above 600 °C, Li₂O enters a relatively stable state, suitable for maintaining lithium source integrity during high-temperature processing.
- This analysis clearly defines the transformation pathway:
LiOH·H₂O → LiOH → Li₂O - The identified critical temperature ranges provide valuable guidance for material formulation, thermal processing, and sintering optimization.
At CeramXpert, we support advanced material research with high-quality thermal analysis consumables designed for accuracy, durability, and compatibility across leading instrument brands.
📩 Feel free to contact us for reliable alternatives and customized solutions for your thermal analysis needs.
#CeramXpert #ThermalAnalysis #STA #TGA #LithiumHydroxide #BatteryMaterials #MaterialsScience #Sintering #NewEnergy #LabConsumables #Ceramics