Hydrogen Storage Material NaAIH4 Dehydrogenation Reaction
Currently, metal hydrides are widely used as hydrogen storage materials in the fuel cell and battery industries for electric vehicles. They are also promising in organic chemistry as strong reducing agents and have potential applications in the hydrogen economy. It is speculated that in the future, hydrogen fuel will replace gasoline and diesel in the automotive sector. Complex hydrides, such as Sodium Aluminum Hydride (NaAlH4) or Lithium Borohydride, are used for hydrogen storage, offering the same hydrogen storage density as methane, without the need for energy input like methane, and releasing hydrogen as free atoms.
Test Conditions:
- Instrument: STA409PG (with steam furnace)
- Temperature Range: Room temperature to 500°C
- Sample Mass: 9.6 mg
- Heating Rate: Isothermal -> Ramp Up
- Crucible: Al2O3 (Alumina)
- Atmosphere: N2/10% H2O, 110 ml/min
- Sensor: S-type TG-DSC sensor
Results and Discussion:
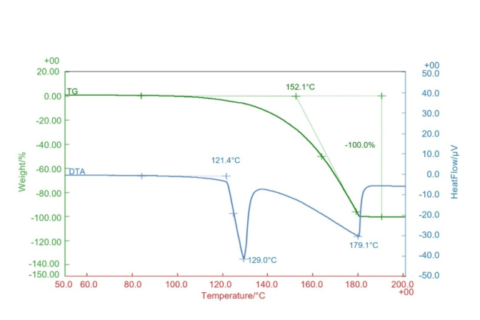
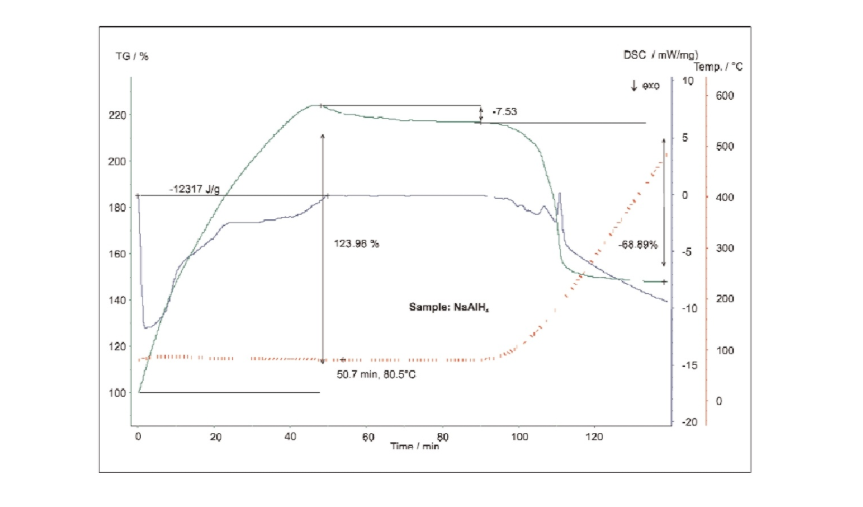
The NaAlH4 sample was tested at a specific humidity using DSC-TG synchronous thermal analysis with a steam furnace. The sample was first held at 80°C, then ramped to 500°C. During the initial phase of testing, NaAlH4 reacted with steam, causing an increase in sample mass, while the corresponding DSC curve showed a strong exothermic peak. This reaction released hydrogen while forming hydroxides. Once the temperature exceeded 100°C, the hydroxides decomposed to form the corresponding oxides. At this stage, the sample exhibited bubbling, and a strong endothermic effect was observed on the DSC curve.
At CeramXpert, we provide high-quality Alumina Crucibles suitable for various thermal analysis applications. These crucibles ensure precise testing and accurate results, especially in hydrogen storage and release studies.